Crude oil
Separated into its different components by fractional distillation
Petroleum gas
Petrol
Naphtha
Kerosene (paraffin)
Diesel
Lubricating oil
Bitumen
Hydrocarbons are compounds made only of hydrogen atoms and carbon atoms.
Separated into its different components by fractional distillation
Petroleum gas
Petrol
Naphtha
Kerosene (paraffin)
Diesel
Lubricating oil
Bitumen
Hydrocarbons are compounds made only of hydrogen atoms and carbon atoms.
Alkanes
Substitution
Propane + chlorine --> chloropropane + HCl
Special conditions: UV light
Propane + chlorine --> chloropropane + HCl
Special conditions: UV light
Alkenes
Addition (halogenation)
Alkene + any halogen --> halogen-alkane
Special conditions: NA
Alkene + any halogen --> halogen-alkane
Special conditions: NA
Addition (hydrogenation)
Alkene + hydrogen --> alkane
Special conditions: 180 deg, Nickel catalyst
Alkene + hydrogen --> alkane
Special conditions: 180 deg, Nickel catalyst
Addition (hydration)
Alkene + steam --> alcohol
Special conditions: 300 deg, 65 atm, propanoic acid catalyst
Alkene + steam --> alcohol
Special conditions: 300 deg, 65 atm, propanoic acid catalyst
Alcohols
Fermentation
Glucose --> ethanol + carbon dioxide
Special conditions: 37 deg, yeast
*To manufacture ethanol, hydration of ethene is preferred over fermentation of glucose because
1. The yeast dies when the concentration of ethanol gets too high
2. The fermentation process takes a few days
Glucose --> ethanol + carbon dioxide
Special conditions: 37 deg, yeast
*To manufacture ethanol, hydration of ethene is preferred over fermentation of glucose because
1. The yeast dies when the concentration of ethanol gets too high
2. The fermentation process takes a few days
Oxidation
Alcohol + any oxidizing agent --> carboxylic acid + water
Alcohol + any oxidizing agent --> carboxylic acid + water
Observations:
If oxidizing agent = potassium manganate (VII),
solution turns from purple to colorless
If oxidizing agent = potassium dichromate (VI)
solution turns from orange to green
If oxidizing agent = potassium manganate (VII),
solution turns from purple to colorless
If oxidizing agent = potassium dichromate (VI)
solution turns from orange to green
Carboxylic acids
All normal acid reactions
Carboxylic acid + metal --> salt + hydrogen
eg. ethanoic acid + sodium --> sodium ethaboate + hydrogen
Carboxylic acid + metal carbonate --> salt + water + carbon dioxide
eg. ethanoic acid + sodium carbonate --> sodium ethaboate + water + CO2
Carboxylic acid + alkali --> salt + water
eg. ethanoic acid + sodium hydroxide --> sodium ethaboate + water
Carboxylic acid + metal --> salt + hydrogen
eg. ethanoic acid + sodium --> sodium ethaboate + hydrogen
Carboxylic acid + metal carbonate --> salt + water + carbon dioxide
eg. ethanoic acid + sodium carbonate --> sodium ethaboate + water + CO2
Carboxylic acid + alkali --> salt + water
eg. ethanoic acid + sodium hydroxide --> sodium ethaboate + water
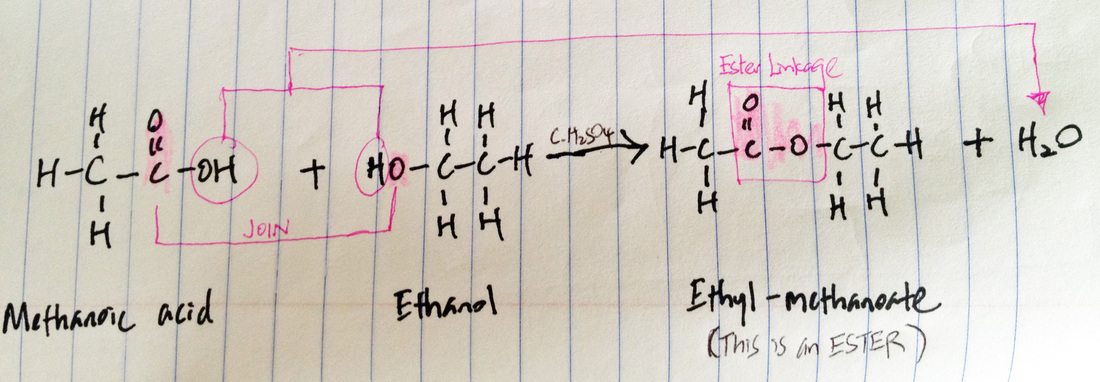
Esterification
Special conditions: reflux, conc. sulfuric acid
Macromolecules
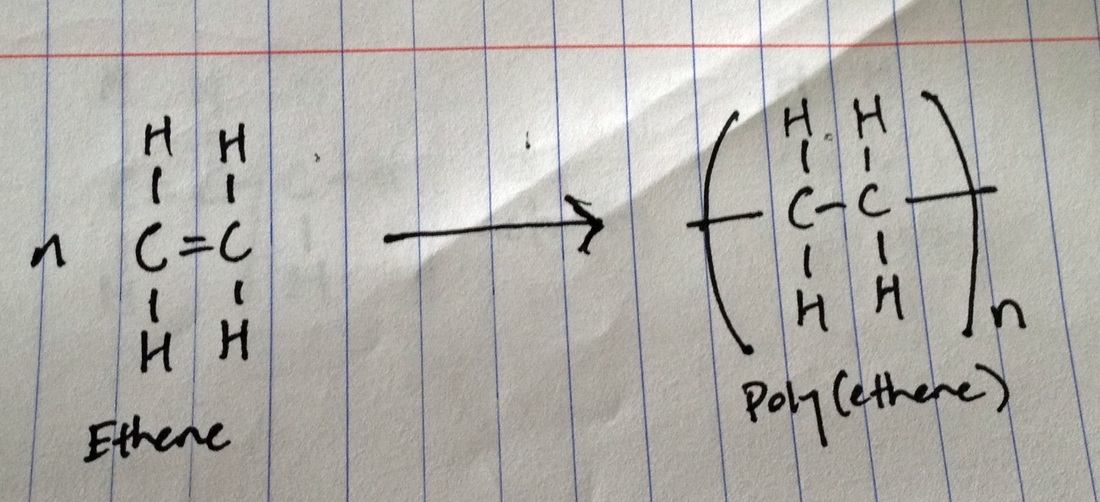
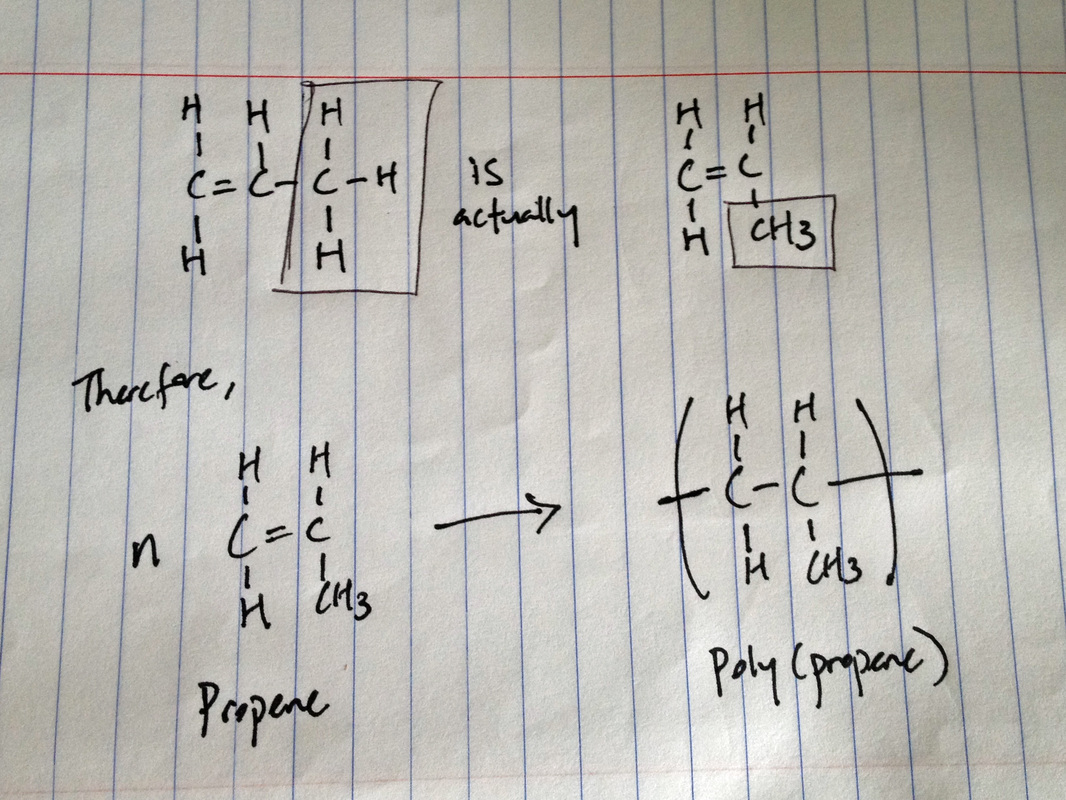
Addition polymerization
n(alkene) --> poly(alkene)
n(alkene) --> poly(alkene)
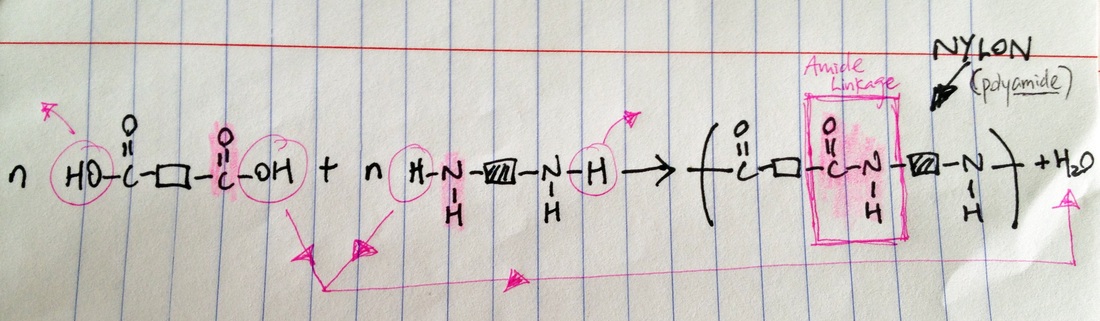
Condensation polymerization






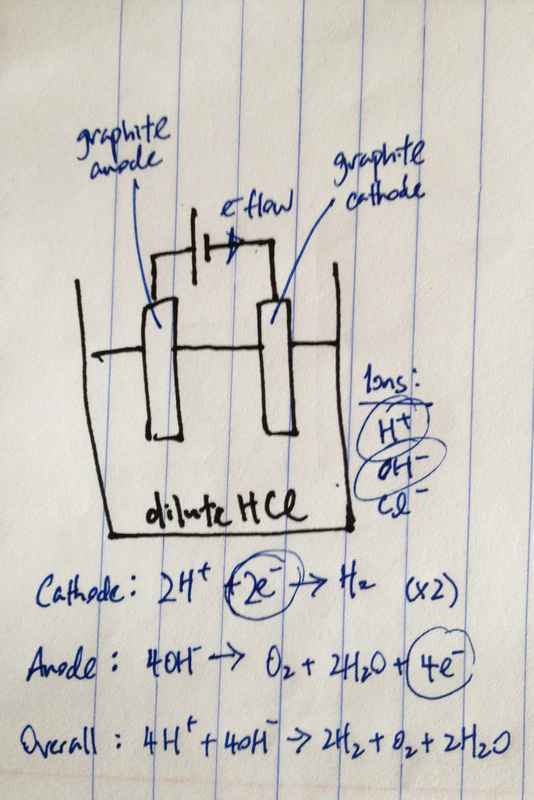

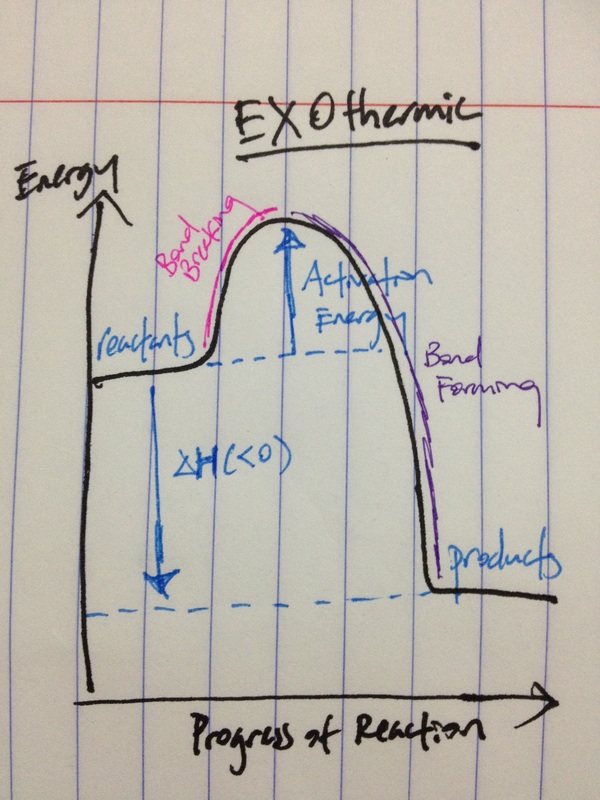
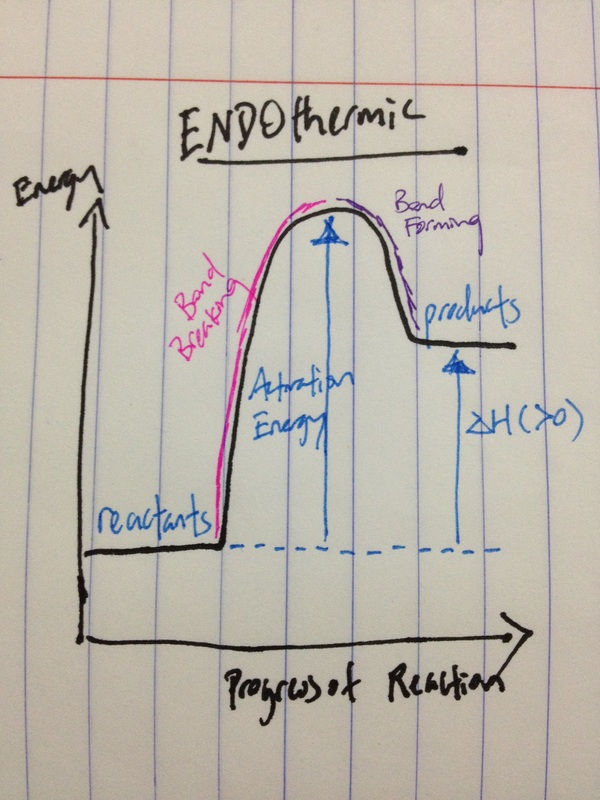
 RSS Feed
RSS Feed